|
As you may know, Sulfur has six valence electrons, and the Fluorine has seven valence electrons.  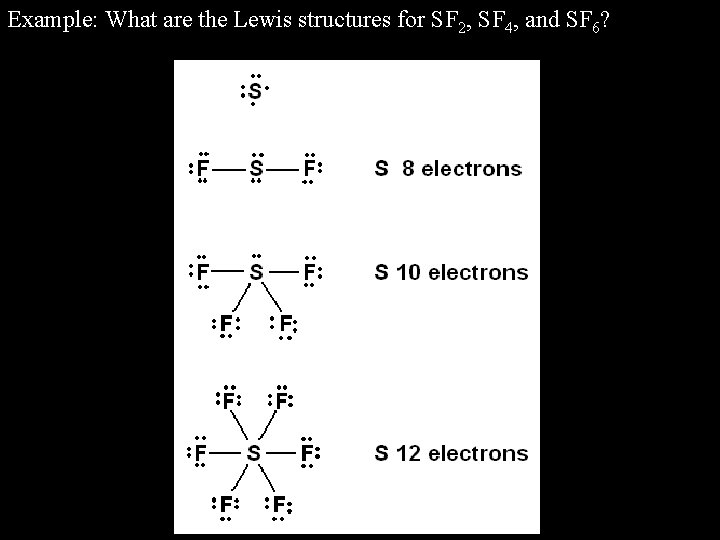
To know about the Sulfur Fluorine - SF2 molecule geometrythe very first thing we have to do is to add up the valence electrons. So in this article, I am going to solve all the confusions regarding of the Sulfur DiFluoride - SF2 molecular geometry. There are so many things to know about such as molecular geometry, Lewis structure, polarity, hybridization, as well as bond angles, but very little information available online.  
Many of my students were confused after not getting any useful information about SF2 on the internet.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
